This is the summary on WHO Considerations in the investigation of cases and clusters of COVID-19 & Coronavirus disease (COVID-19) technical guidance: Early investigations protocols
Objective of the investigation
- -Rapidly detect COVID-19 and any evidence of human-to-human transmission among contacts
- -Reduce human –to-human transmission, prevent outbreaks, and delay the spread of disease
Composition, protection of and tools for the investigative team
Composition
- Personnel with the capacity, knowledge, and authority to:
- -Interview persons with suspected COVID-19 and conduct contact tracing
- -Triage suspected COVID-19 cases and contacts for health care depending on clinical condition
- -Collect respiratory specimens from suspected COVID-19 cases
- -Recommend and implement measures to prevent further transmission
Protection
- The COVID-19 virus is spread through contact, droplet, and fomites. To minimize risk of infection of the investigation team:
- -Optimize size of team to minimize contact with a suspected COVID-19 case
- -Ensure all those in the investigation team are trained in IPC measures specific to COVID-191
- -Interview suspected cases and contacts over the phone, if feasible, or at a distance of more than 1 meter.
Tools
- -Provide sufficient and appropriate PPE2
- -Gather biological specimen collection material, transport containers, viral transport media, labels, bags, coolers, and cold packs
- -Gather copies of case investigation protocols, questionnaires, contact tracing and monitoring tools, and the national case definition
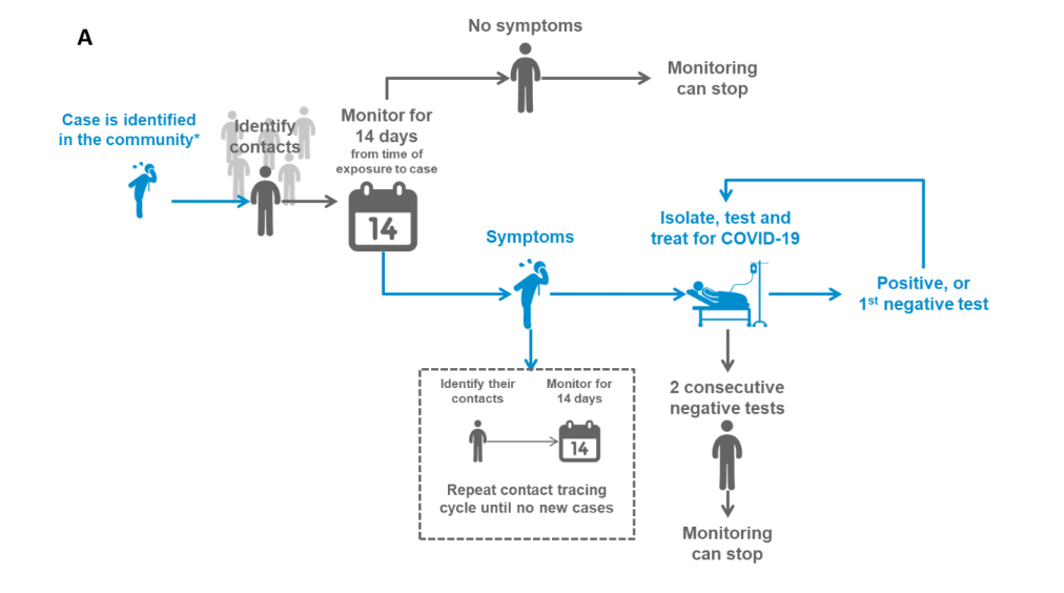
Contact Tracing
Identify all social, familial, work, and health care worker contacts who have had contact with a confirmed case from 2 days before symptom onset of the case and up to 14 days after their symptom onset. Create a line list, including demographic information, date of first and last common exposure or date of contact with the confirmed or probable case, and date of onset if fever or respiratory symptoms develop.
Reporting
National authorities need to report probable and confirmed cases of COVID-19 to WHO within 48 hours of identification. Report using the Individual Case Reporting Form and consider transitioning to the aggregate Daily/Weekly Reporting Form if the number of cases increases and resources are no longer available for individual case reporting.
Further Investigation
Standardized epidemiological protocols have been developed for COVID-19 and are available on the WHO website. These may be initiated in addition to the public health investigation, but should not replace the investigation:
- -First few cases and contacts transmission investigation protocol, which evaluates extent of infection among cases and their contacts
- -Household transmission investigation protocol, which evaluates extent of infection within the household setting
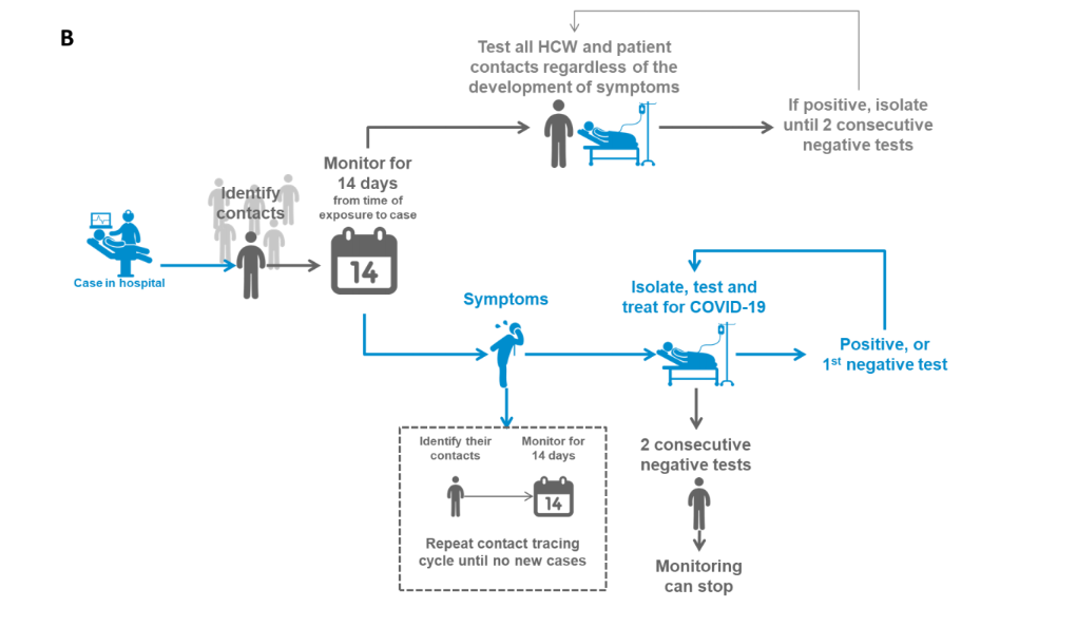
- -Assessment of risk factors for COVID-19 among health care workers working in a health care setting in which a confirmed COVID-19 patient is receiving care
- -Surface sampling of COVID-19 virus: A practical ‘how to’ protocol for health care and public health professionals to assess surface contamination and the role of environmental contamination in transmission
- -Global COVID-19 Clinical Characterization Case Record Form and data platform for anonymized COVID-19 clinical data to collect clinical data to better understand the natural history of disease and describe clinical phenotypes and treatment interventions
Early Investigation
Several early investigation master protocols or master forms for COVID-19 are available for countries. They are summarized below:
The First Few X (FFX) Cases and contact investigation protocol for COVID-19 Infection
This protocol has been made by WHO for cases and contacts in the general population or can be used in restricted to close settings (like households, healthcare settings, school). This is specifically to investigate the First Few X number of cases and their close contacts (FFX). By this, epidemiological exposure data and biological samples can be systematically collected and shared rapidly in a format that can be easily aggregated, tabulated and analyzed across many different settings globally for timely estimates of COVID-19 infection.
Household transmission investigation protocol for COVID-19 infection
This investigation is intended to provide rapid and early information on the clinical, epidemiological and virological characteristics of COVID-

Protocol for assessment of potential risk factors for COVID-19 infection among health care workers in a health care settings
The following protocol has been designed to investigate the extent of infection and risk factors for infection among health care workers. Follow-up and testing of respiratory specimens and serum of health care workers within a facility in which a confirmed case of COVID-19 infection is receiving care can provide useful information on transmissibility and routes of transmission and are important for limiting amplification events in health care facilities.
Population-based age-stratified seroepidemiological investigation protocol for COVID-19 virus infection
This is a population-based, age-stratified prospective study protocol to assess COVID-19 virus infection. It is intended to provide key epidemiological and serologic characteristics of COVID-19 in general population including the extent of infection in the general population and age-specific infection cumulative incidence, which is determined by seropositivity, and the fraction of asymptomatic or subclinical infection.

Surface sampling of coronavirus disease COVID-19 virus: A practical “how to” protocol for health care and public health professionals
This pandemic was at first has no clear role of environmental contamination in transmission of the disease. This protocol was designed to determine virus presence and persistence on fomites in various locations where a patient infected with COVID-19 is currently receiving care or being isolated, and also to understand how this may relate to COVID-19 transmission events in these settings.
